Comparison of NRTI combos
AMBER Study: D/C/F/TAF QD vs D/C + F/TDF QD
Original article :
Eron J, EACS 2017, Abs. PS8/2
Last update :
22/12/2017
Dr Anton Pozniak
Chelsea and Westminster Hospital
London, UK
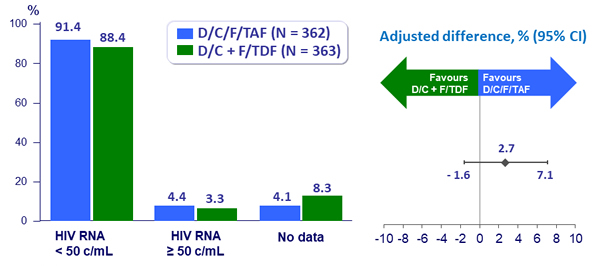
- Through Week 48, D/C/F/TAF resulted in high virologic suppression in treatment-naïve patients (91.4% ; FDA Snapshot) that was non-inferior to DRV/C + F/TDF (88.4%)
- No development of DRV, primary PI or TDF/TAF RAMs
- Onepatient developed M184I/V (D/C/F/TAF arm)
- Few serious adverse events, grade 3/4 adverse events, or adverse events-related discontinuations
- No discontinuations due to bone, renal, or CNS adverse events
- Bone, renal and lipid safety consistent with known profiles of TAF and cobicistat
Design
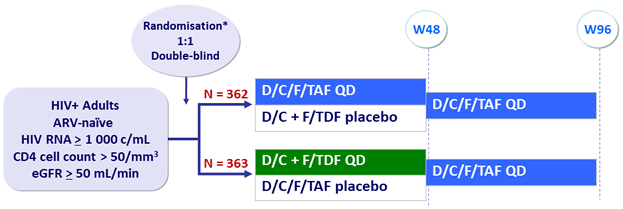
*
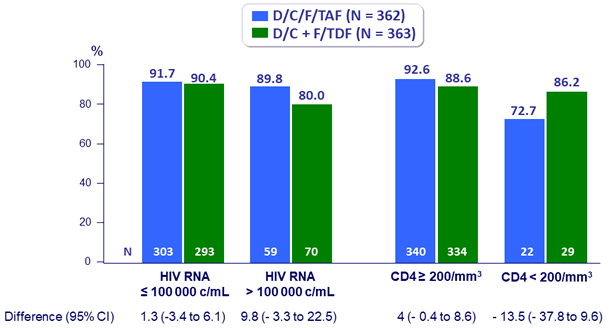
Randomisation was stratified by HIV RNA (< or ≥ 100 000 c/mL) and CD4 cell count (< or ≥ 200/mm3)
Objective
- Non inferiority of E/C/F/TAF at W48: % HIV RNA < 50 c/mL by intention to treat, snapshot analysis (lower margin of the 95% CI for the difference = -10%)
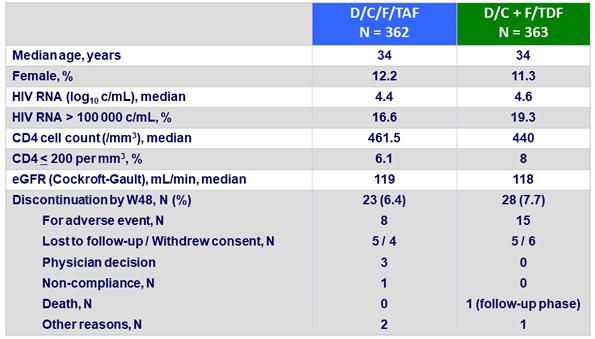
Baseline characteristics and patient disposition
Virologic outcome at W48 (ITT, snapshot)
Resistance analysis
- Virologic failures with paired genotypes (baseline and failure with HIV
RNA ≥ 400 c/mL), N = 9 [7 D/C/F/TAF + 2 D/C + F + TDF]:
1 patient with emergence of M184I/V (D/C/F/TAF)
Virologic outcome at W48 by baseline strata (ITT, snapshot)
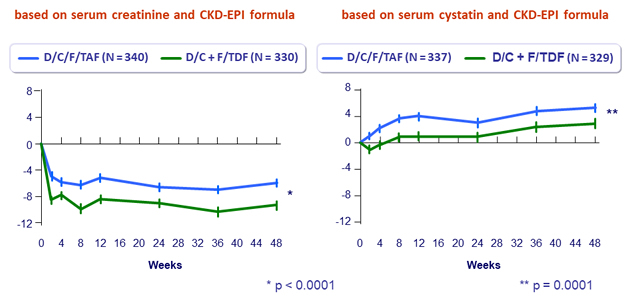
Mean (SE) change from baseline in eGFR (mL/min/1.73m²)
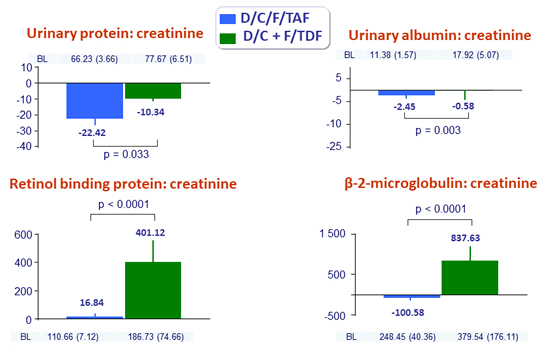
Mean (SE) changes (mg/g) in proteinuria at W48
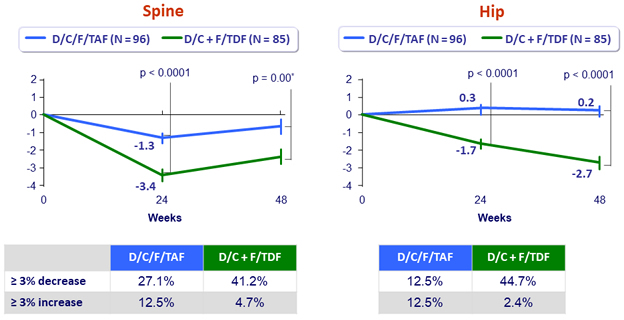
Mean (SE) % changes in bone mineral density at W48
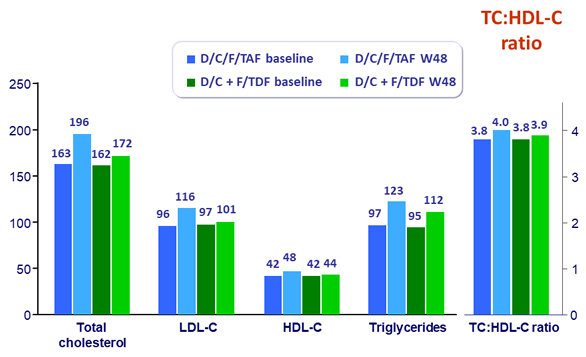
Median lipid values (mg/dL)
Adverse events through W48, %
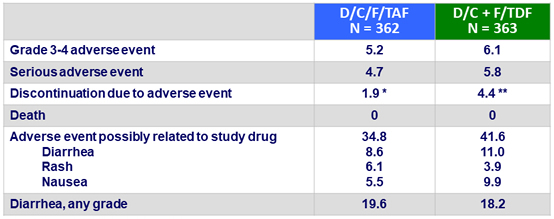
* rash (N = 6); diarrhea (N = 1)
** rash/erythema (N = 7), diarrhea (N = 1), toxic skin eruption (N = 2), Stevens Johnson syndrome (N = 1), bone marrow oedema (N = 1), increased beta-2 macroglobulin (N = 1), arthralgia (N = 1), neoplasm (N = 2)
- No grade 3 or 4 laboratory abnormalities in ≥ 5% of patients in either arm
- No discontinuation of D/C/F/TAF for bone, renal or CNS adverse event
 Back to Table of Contents
Back to Table of Contents



