Baseline characteristics and patient disposition
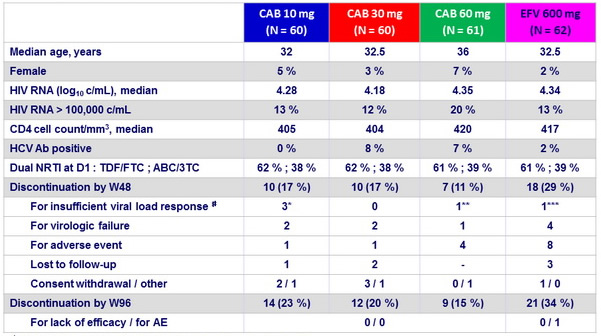
# W20 HIV RNA : * : 51, 107, 189 c/ mL ; ** : 108 c/ mL ; *** : 146 c/ mL
HIV RNA < 50 c/ml (ITT-E, snapshot )
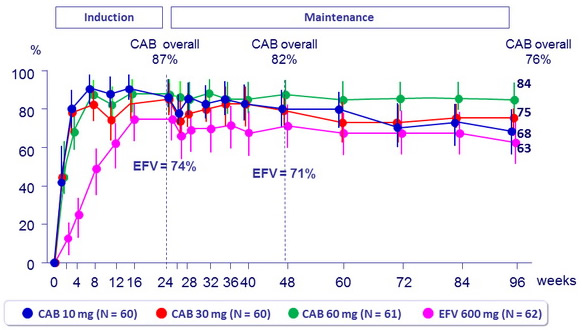
Week 96 outcome, ITT -ME , snaphsot
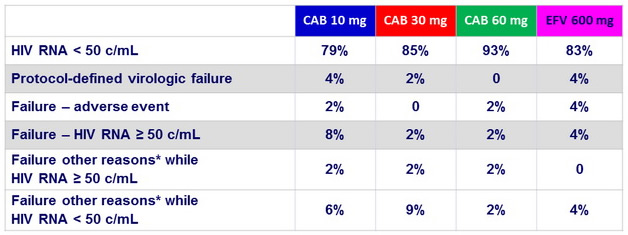
* Other reasons : missing data, protocol deviation, non-compliance, lost to follow-up, withdrawn consent, investigator discretion, ART change, ineligible for maintenance phase
Protocol-defined virologic failure (PDVF)
- Non-response: < 1 log 10 c/mL decrease of HIV RNA by Week 4, unless
< 400 c/mL ; or HIV RNA ≥ 200 c/mL on or after Week 16
- Rebound: HIV RNA ≥ 200 c/mL after confirmed < 200 c/mL ; or > 0.5 log 10 c/mL above nadir (the lowest prior HIV RNA ≥ 200 c/mL )
- Both non-response and rebound required consecutive confirmatory results
Protocol-defined virologic failures
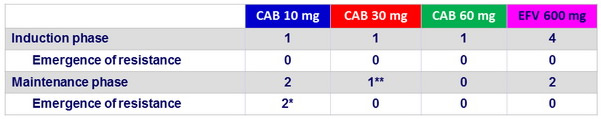
* CAB 10 mg : emergence of NNRTI (E138Q) and INI (Q148R) mutations at W48; CAB FC = 3, RPV FC = 2 ; CAB 10 mg : emergence of NNRTI mutations (K101K/E + E138E/A) but not to INI
Additional patient on CAB 10 mg : failure at W48 not confirmed , mutation to NNRTI (K101K/E + E138E/K), no INI mutation
** CAB 30 mg : PDVF at W36 with no emergence of NNRTI mutation ( integrase not amplified )
Adverse events
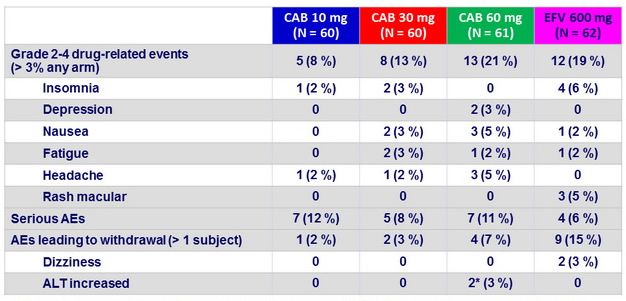
* 2 subjects with steatohepatitis developed asymptomatic grade 4 ALT elevations (meeting protocol-defined liver stopping criteria ) with normal bilirubin levels , at W4 and W8, which resolved off investigations products.
Laboratory abnormalities
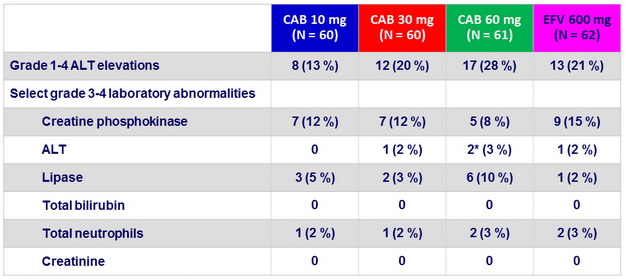 * 2 subjects with steatohepatitis developed asymptomatic grade 4 ALT elevations (meeting protocol-defined liver stopping criteria ) with normal bilirubin levels , at W8, which resolved off investigations products.
* 2 subjects with steatohepatitis developed asymptomatic grade 4 ALT elevations (meeting protocol-defined liver stopping criteria ) with normal bilirubin levels , at W8, which resolved off investigations products.



